|
Due to the presence of equal number of negative electrons and positive protons, the atom on the whole is electrically neutral. Electrons have negative charge, protons have positive charge whereas neutrons have no charge. Due to a technicality, however, Einstein was awarded his in 1922 instead of 1921. 1) An atom is made up of three particles: electrons, protons and neutrons. Bohr received his for his atomic model while Einstein’s was for his work on the photoelectric effect. Niels Bohr in front of the University of Cambridge. Young Niels Bohr- by AB Lagrelius & Westphal- Wikimedia Commons. In fact Nield found that the ratio of energy in electrons and the frequency of their orbits around the nucleus was equal to Planck's constant (the proportion of light's energy to its wave frequency, or approximately 6.626 x 10-23 ). Neils Bohr wasn’t as good a writer as he was a student in his younger years. Thus the model simply throws out classical electromagnetic theory. When an electron is in an 'allowed' orbit it does not radiate. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. The model asserts that: The planetary model is correct. This approach allowed Niels to explain a relatively stable atomic Model, which still hinged on the orbital model that Rutherford postulated. Then in 1913 Bohr, by accident, stumbled across Balmers numerology for the hydrogen spectrum, and in a flash came up with a workable model of the atom. Niels Bohr which after his PhD circa 1912 had joined Rutherford realized that Rutherford's model wasn't quite right and started to articulate a model that borrowed from Planck's quantum theory. Niels Bohr was a Nobel Prize-winning physicist and humanitarian whose revolutionary theories on atomic structures helped shape research worldwide. All of the negative charge was held in the electrons, orbiting the dense nucleus like planets around the sun. This model depicted an atomic model with nearly all its mass, and positive charge, in a central nucleus about 10,000 times smaller than the atom itself. Rutherford experiment with alpha particles shot at a thin gold foil resulted in the Rutherford model of the atom (Orbital Model). Neils Bohrs 'New' Model for the anatomy of the atom, while flawed, was one of the most important intellectual steps in human understanding of the universe. Thomson actually) which blew a big hole in "Plum Pudding Hypothesis" of the atom.  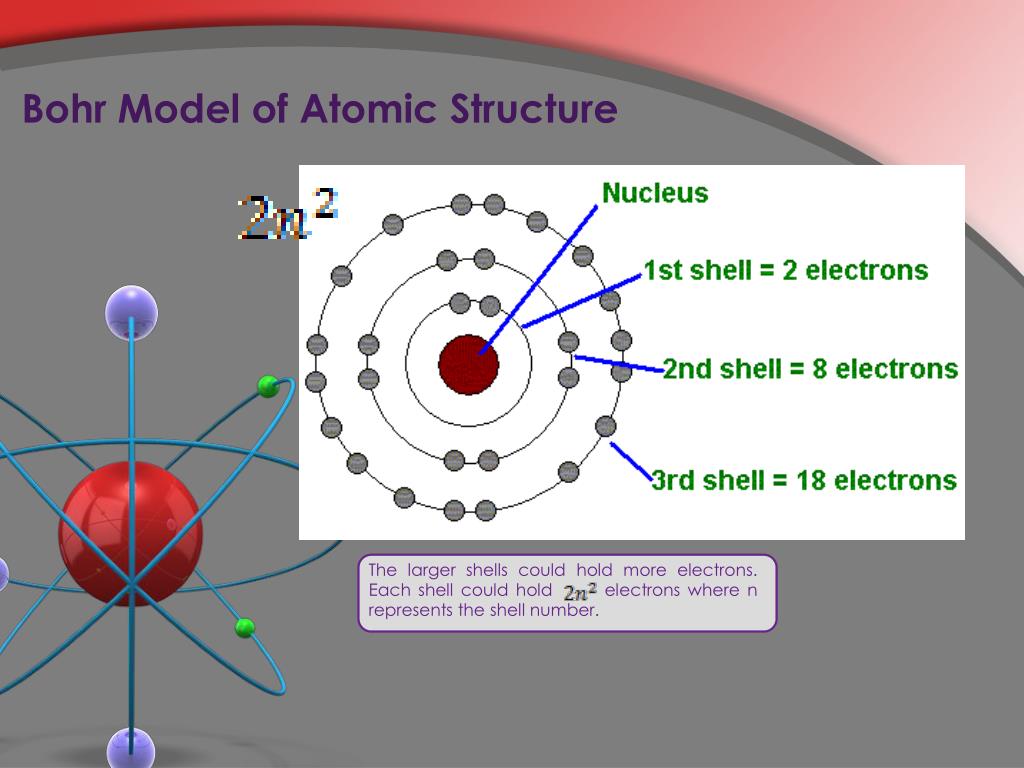
Thomson that resulted "Plum Pudding" model of the atom and the 2nd one by Rutherford (a student of J.J. Well there were two experiments back to back one by J.J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
